
FDA on Prefilled Syringes and Combination Products — What This Means for You – Quality by Design for Biotech, Pharmaceutical and Medical Devices

FDA Releases Guidance for Instructing Patients in Self-Swabbing for SARS-CoV-2 Testing - Clinical Advisor

FDA Guidance Needed to Assure Safe Labeling Practices by 503A and 503B Compounders | Institute For Safe Medication Practices

The FDA issues final guidance for emerging post-market medical device signals - PharSafer® - Specialists in Global Clinical and Post Marketing Drug Safety
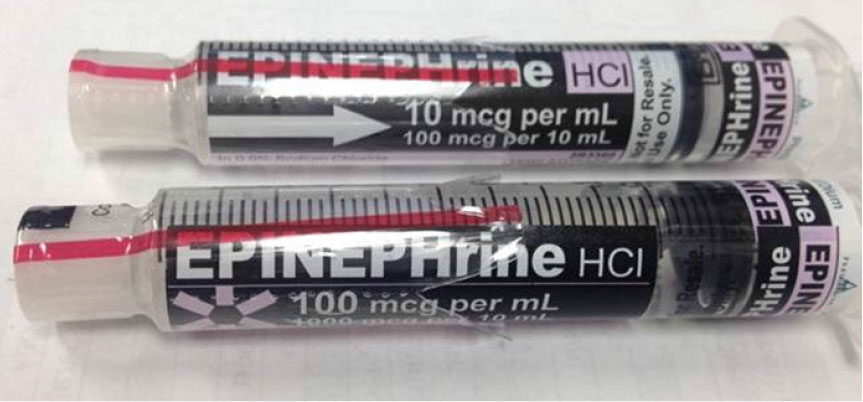
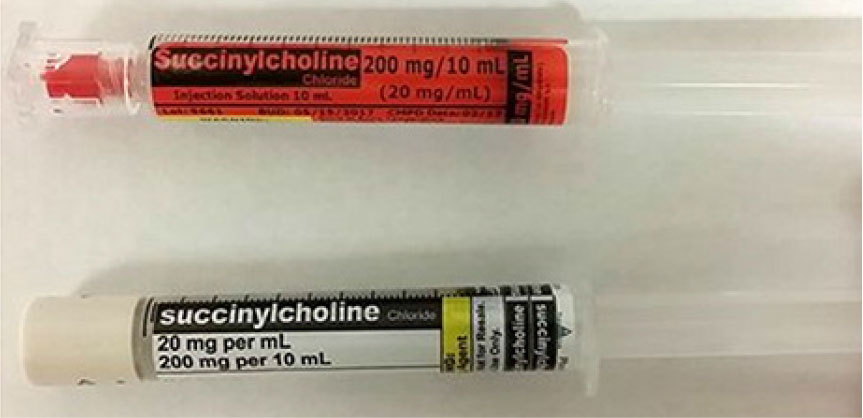
FDA Guidance Needed to Assure Safe Labeling Practices by 503A and 503B Compounders | Institute For Safe Medication Practices

FDA Draft Guidance Clarifies Evidentiary Standards for Interchangeable Biological Products - Biosimilars







/cdn.vox-cdn.com/uploads/chorus_asset/file/24599426/1244633034.jpg)

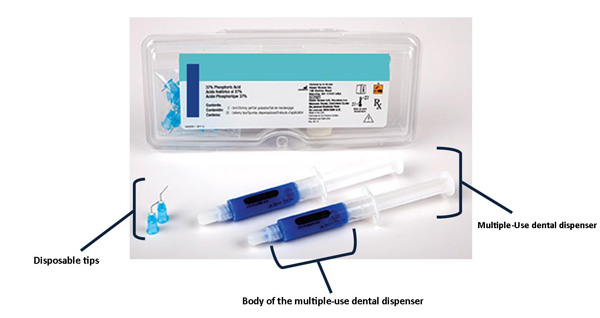




![Best Syringes for Laboratory Use [Buying Guide] | Air-Tite Products Co Best Syringes for Laboratory Use [Buying Guide] | Air-Tite Products Co](https://www.air-tite-shop.com/images/product/large/272.jpg)
